The importance of BioBanking
Biobanking has become an important factor in research, supporting mainly genomic and personalized research. Biobanks allow researchers access to data representing a large number of people. Samples in biobanks and the data derived from those samples can often be used by multiple researchers for cross purpose research studies. The advantage of biobanks is huge as it opens an immense amount of shared samples for researchers to use when researchers struggled to acquire sufficient samples.
Running a Biobank requires adhering to high standards of privacy and confidentiality, in addition to maintaining high standards of data integrity and inventory management. Biobanks must store the samples at the highest standards of order, security and maintenance. Samples are registered in a sample management system that allows full documentation, recording and registering every required detail about each sample, noting the physical location of each sample to enable faster location of specimens.
and registering every required detail about each sample, noting the physical location of each sample to enable faster location of specimens.
High-quality samples are crucial for research and require maintaining and monitoring them in the highest standards all the way- storage location, chain of custody, experimental data and so on.
Managing samples is a major challenge for research labs in general and biobanks in particular. Labs can produce and manipulate a large number of aliquots every day. Organizing them might be overwhelming and time-consuming, and prone to human errors. Using a lab manager software to set up bio collections helps biobanks manage samples efficiently and facilitates collaboration among lab mates and collaborators.
Importing your collection into a sample management system
One of the first steps you need to take to create a well-planned sample management is populating your inventory collections. When looking for a sample management system it is important to make sure you can import your collection items in bulk to save you time and avoid human errors. Also, it is advised to set in advance your collections and the specific characteristics of your samples to easily import and upload all your samples easilyת orderly and efficiently.
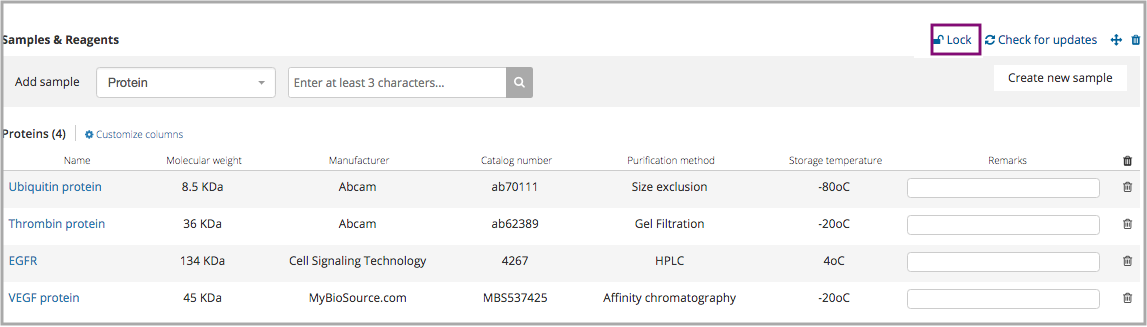
Locking sample elements in protocols
To maintain data integrity, have full control of your data, and manage your research processes, it is advised to create structured protocols and have the option to lock sample elements that were added to a section. Locking the samples in a certain protocol limits the ability of collaborators to change the selection of the samples, and it only allows them to choose the individual stocks.
Adding samples to experiments
Having the ability to add samples to experiments will allow you to document your work easily and efficiently, within minutes, by adding samples directly to your experiment, eliminating the need to actively open storage cabinets. Additionally, track sample usage directly in your inventory notebook., by linking it to your experiments. Adding samples to experiments will help you maintain context between your experiments and inventory.
Linking your samples to your experimental procedures allows you to fully record every single step performed in the lab work. Not only do you have a complete record of which samples were used, including each individual stock, but also an update on real-time of how much sample is left in the tube. The record of the sample usage is now linked to each specific experiment in a bidirectional way, so you have a complete history of where, how and when every single sample was used.
This comes to solve traceability, data integrity, and history to be able to further reproduce the experimental work as well as submitting it for the regulation purposes.
Connecting results to samples
By connecting actual results from data files from experiments to your samples you will gain a great overview of all the data that was collected in your lab on a specific item. This can be done by using datasets. For example, when you add an attachment as a Dataset, Labguru will then scan the dataset and locate all inventory items that appear in it. It will then add the relevant data from the file and will display it in a new tab in the item’s page (named ‘Datasets’).
Documenting produced samples
Labguru lab manager software makes a step further in helping researchers organise their data and maintain chain of custody. With a sample management system like Labguru, you can log and document all the data related to a certain sample, maintaining data integrity and good clinical laboratory practices. Each sample history has a separate log and according to 21CFR#11. Labguru maintains an audit trail (https://my.labguru.com/audit_trail) to all system records – every change to the system is logged in a ‘histories’ table – thus confirming the requirement of maintaining an audit trail. Each audit-trail entry is time-stamped – we record the data transferred to the system and the user sending the request. In addition, using Labguru inventory management module will allow you to not only know where the boxes are but rather what is INSIDE them and what is the status of each sample.
To learn more about how Labguru can help you manage your samples


%20(4).png)