Key benefits
Manage Pharmaceutical Storage and Equipment
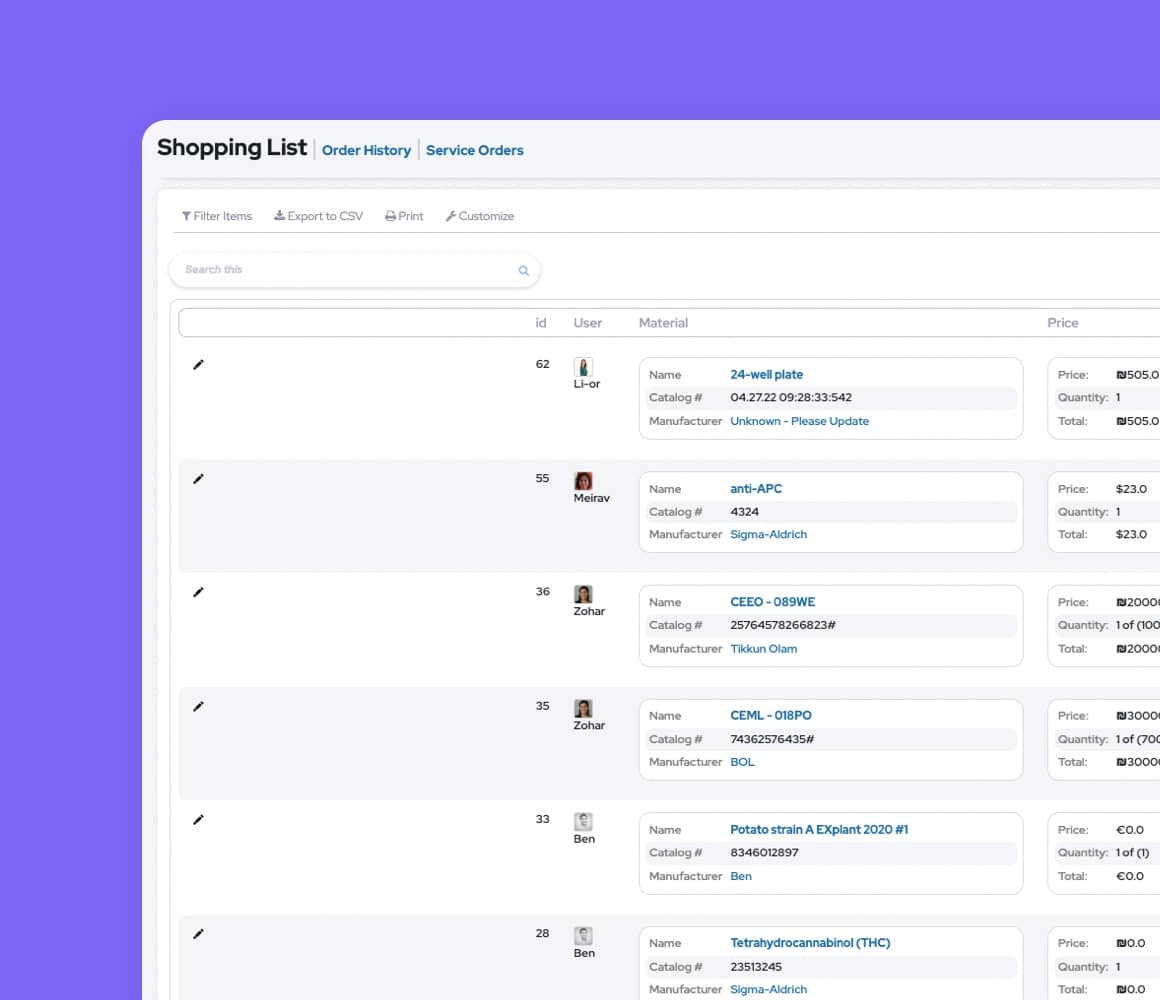
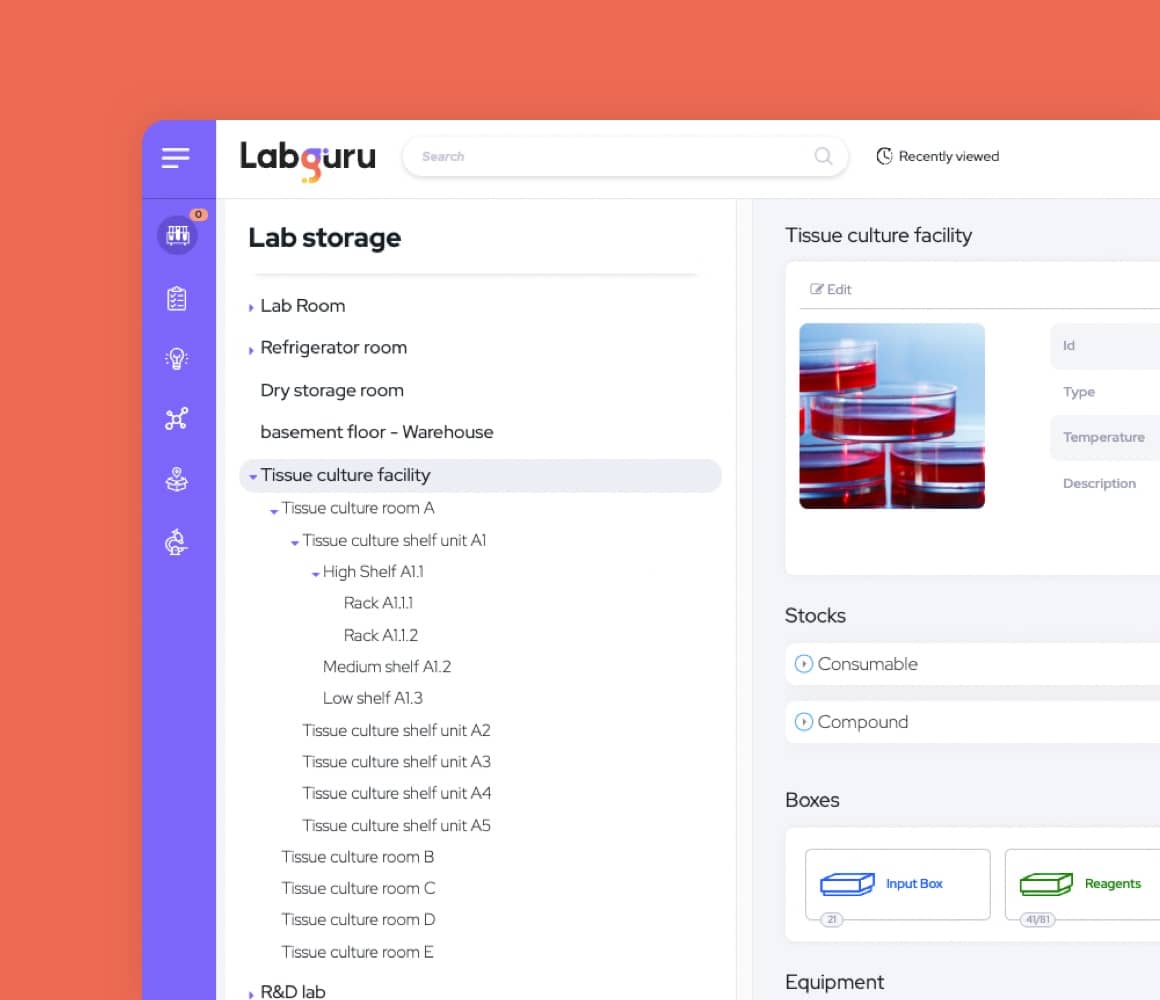
Use Labguru's Pharma LIMS to track the location, expiration date, and remaining quantity of each supply in your lab. Manage and approve pharma shopping lists in a centralized manner to prevent duplicate orders and reduce lab costs. Manage pharmaceutical equipment calibration and schedule equipment usage sessions.
Track Your Samples and Collections
Manage collections such as antibodies, plasmids, rodents, cell lines, etc. Use molecular biology and chemistry tools to sketch items or import sketches. Map the lineage of parent-child relationships between items and link samples to experiments. Get real-time updates on remaining stock amounts.
Automate Pharma Research Data Management Processes
Use Labguru Workflows to create step-by-step processes that occur periodically or as a result of actions in the system. Integrate with lab instruments and automatically transfer results to your pharma research management system. Process data and run automated analysis workflows. Automate the creation of reports and certificates.
Enhance Communication & Collaboration
Engage with team members in one centralized system. Discuss experimental data within context and assign tasks seamlessly. Visualize results together and annotate images to leave comments. Collaborate with external scientists and export reports and images easily to facilitate the next breakthrough.
The All-in-One Solution for Pharmaceutical R&D
Labguru combines the best ELN with the best lab management system and advanced informatics.
The platform functions as a central database of samples that is easily accessible to all lab members at all times and from any device connected to the internet.
Save Time, Increase Efficiency
Labguru pharmaceutical LIMS software digitizes and streamlines day-to-day tasks such as data analysis and inventory, equipment, and research data management, saving precious time that would otherwise be wasted on repetitive manual work. We help you do more science: focus on drug development, identify and advance novel leads faster and shorten the time to market.
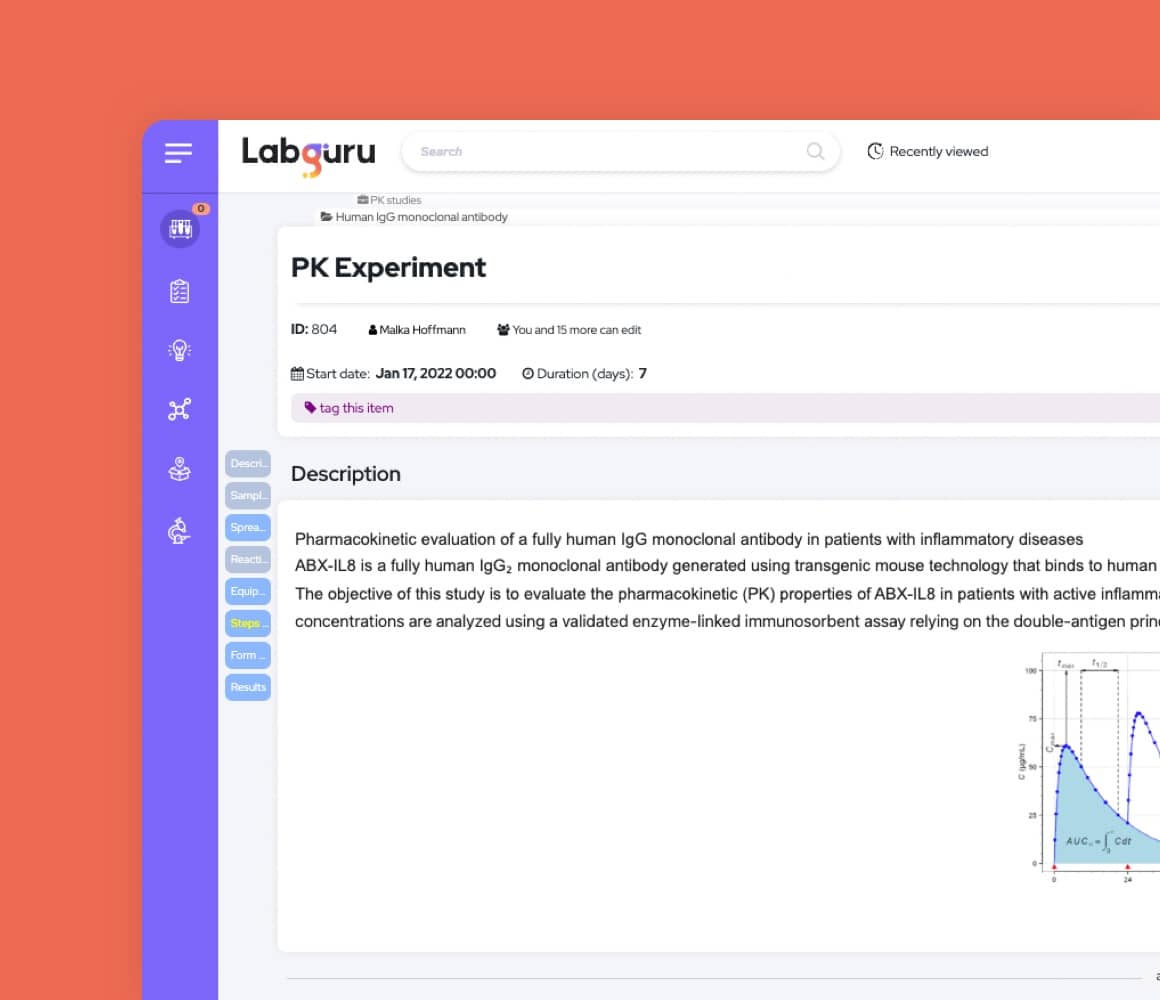
Structure and Centralize Your Data
Labguru assembles all your information in one easily searchable, accessible, and compliant database.
- Organize information in projects and folders
- Bring Labguru with you to the bench and record experiments step-by-step
- Use protocols with reusable forms to make your experiments structured and reproducible
- Access and search historical information
- Annotate results and link them to experiments
- Create reports, compare results using serial datasets, and run analysis queries

"The Labguru team’s experience with supporting academic groups, together with the cloud-based Labguru installation, allows AstraZeneca scientists greater opportunities to share our biological research reagents with external partners"
.png?width=90&height=90&name=lorenz_mayr%20(1).png)
Lorenz Mayr
VP of Reagents & Assay Development, AstraZeneca

Data Integrity and Compliance
We make it easy to comply with regulatory requirements and ensure the accuracy of results.
- Easily create reports or even automate report creation for IND submissions or compliance regulators
- Ensure result accuracy by tracking instrument calibration and receiving notifications when calibration is due
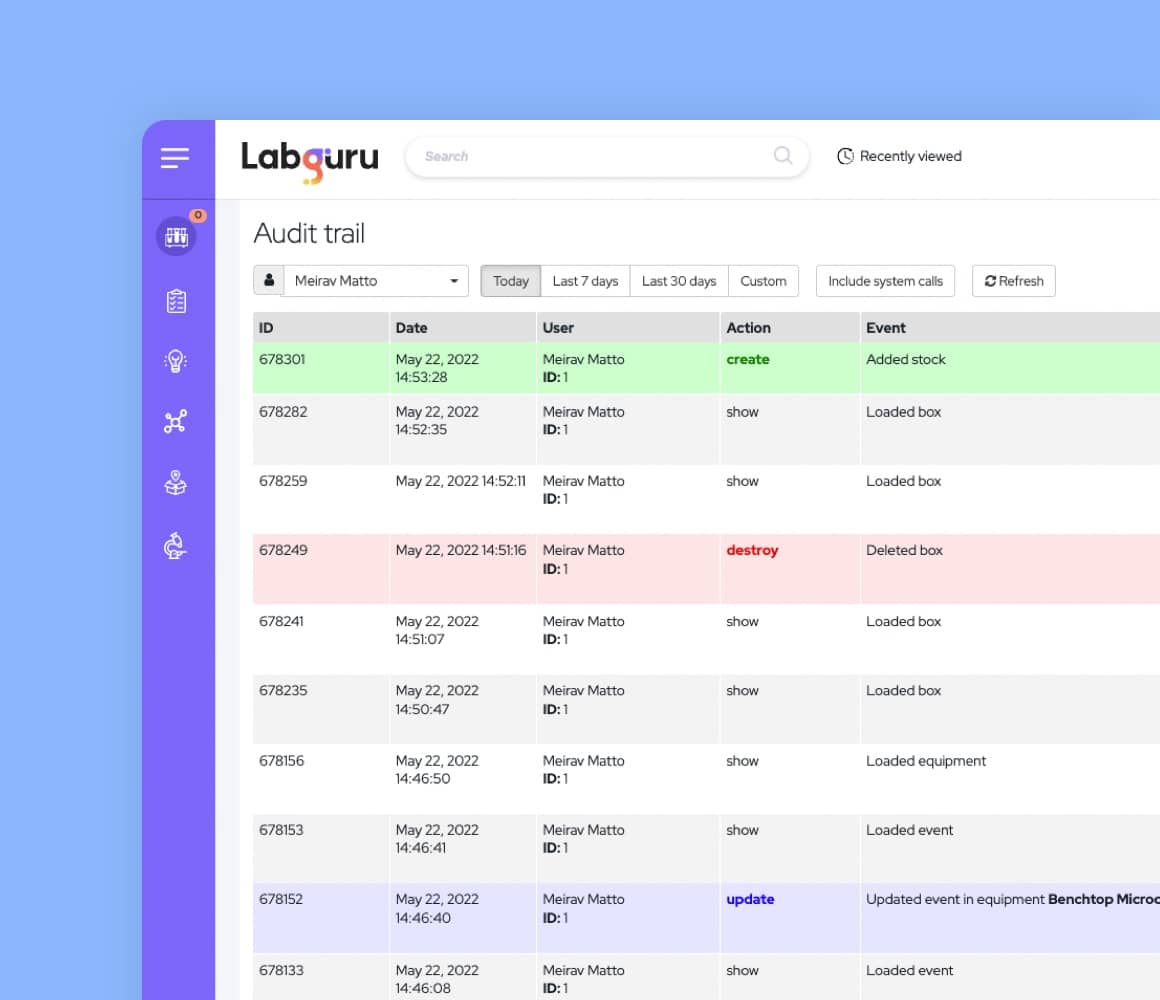
- Full audit trail: all actions in the research management system are recorded and time-stamped with the date, time, user, and action.
- Version history: all historical versions of files, experiments, protocols, etc, are available in PDF form and listed by date
- Sign and Witness: Labguru provides time-stamped digital signing and locking of experiments.
- Link samples and supplies to experiments, to track the exact processes performed on each sample or batch
- Ensure traceability: trace results back to different stages of the drug development process
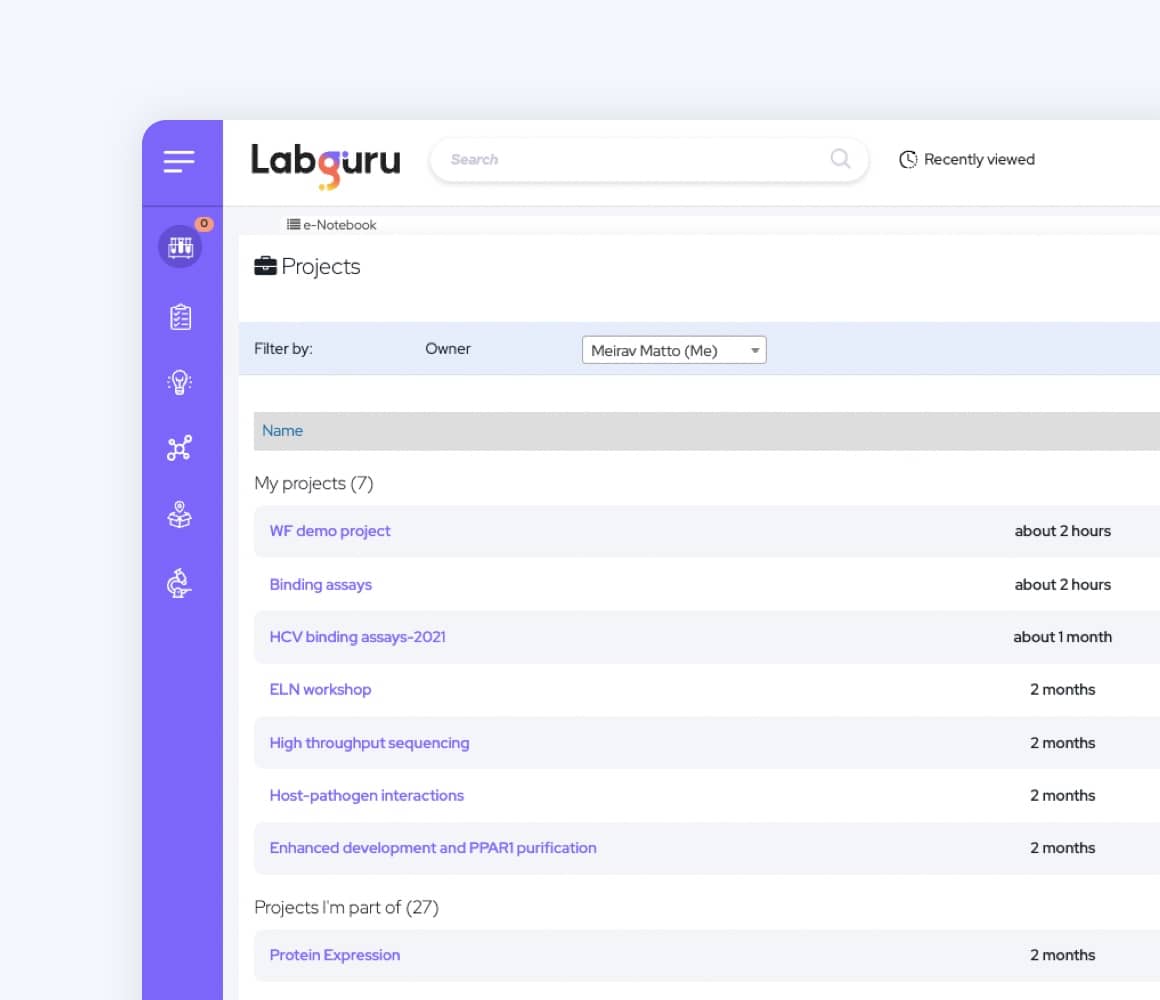
Manage Teams and Projects
Labguru enables smooth collaboration and communication where it matters most.
- Use Gantt charts to plan and monitor the progress of your projects
- Build custom Dashboards to track experiment results, equipment usage, inventory and sample quantities, team milestones, lab purchases, and more
- Run pre-made or custom analysis queries and visualize results in graphs and tables to receive BI and make data-driven decisions
- Assign tasks to lab members
- Create a centralized database for protocols and SOPs to standardize work methods
- Easily create reports and share information
- Create shared experiments for teammates to work on together.
Best Data Security
We leverage best industry practices and security measures to keep your IP safe from competitors and prevent data loss.
- Labguru is ISO 27001 certified and runs on the AWS platform which is ISO 27001, SOC reports 1, 2, and 3 certified.
- We employ multiple layers of protection (Firewalls, traffic monitoring measures, and additional technologies) to always limit access to the Labguru platform to approved networks and users only.
- Data sent over the world wide web to Labguru, including through Transport Layer Encryption (TLS), leverages at least 2048-bit RSA server certificates and 128-bit symmetric encryption keys.
- User information is backed up daily with a retention period of 30 days.
- We maintain a continuously tested data recovery methodology that ensures quick and optimal data retrieval in a crisis scenario.
Why Labguru?
We are an all-in-one solution
Perform efficient assays and maximize your research output. Plan and document experiments, track progress, streamline lab logistics, and share results.
We support you from research to production
Streamline everything from experiment planning to large-scale production on one centralized platform employing advanced technological tools.
We're here to meet your needs
We'd love to learn more about your unique needs to create a tailored solution for your lab. Just let us know how we can streamline your research.
Our solution is intuitive and easy to use
Forget about lengthy onboarding processes and training sessions. Labguru is designed to be so intuitive, you can pick up where you left off.
"The number of samples that can be analyzed is vastly increased as the lab team can write the experiments and results in less than an hour, so I estimate that we save a day per week by using Labguru."
.png?width=90&height=90&name=Bitmap%20Copy%20(1).png)
Charlie Britten
Senior Scientist at ADC Therapeutics
.png?width=120&height=60&name=ADC-Therapeutics-logo%20(1).png)


